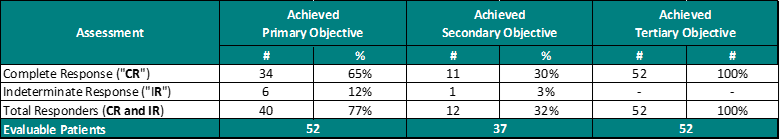
For the primary objective, 65% of Evaluable Patients (Patients evaluated by a Principal Investigator (“PI”)) achieved a Complete Response (“CR”) at any assessment date post primary Study Treatment and 30% of Evaluable Patients achieved a CR at 450 days.
Toronto, May 10, 2023 (ACCESWIRE) – Theralase® Technologies Inc. (“Theralase®” or the “Company”) (TSXV: TLT) (OTCQB: TLTFF), a clinical stage pharmaceutical company focused on the research and development of light activated Photo Dynamic Compounds (“ PDCs”) and their associated drug formulations, used to safely and effectively destroy various cancers, bacteria and viruses, announced today an update on its recently reported Phase II clinical study (“Study II”) interim clinical data.
Background:
Study II was designed to treat patients diagnosed with Bacillus Calmette-Guérin (“BCG”)-Unresponsive, Non-Muscle Invasive Bladder Cancer (“NMIBC”) Carcinoma In-Situ (“CIS”) (with or without resected papillary Ta/T1 disease) with a patented investigational study drug Ruvidar™ (TLD-1433 – a ruthenium based PDC intravesically instilled into a patient’s bladder), subsequently activated by a proprietary investigational study device (TLC-3200 Medical Laser System – a green (520 nm) laser system equipped with fiber-optic light emitters and detectors).
In recent discussions with the Medical and Scientific Advisory Board (“MSAB”) for Study II, the MSAB advised the Company to review the FDA Guidance to Industry1 on how to best classify Indeterminate Response (“IR”) patients (patients assessed with negative cystoscopy and positive urine cytology), where the source of the positive urine cytology has not been determined.
The FDA Guidance to Industry1 states as follow:
“For single-arm trials of patients with BCG-unresponsive disease, the FDA defines a complete response as at least one of the following:
• Negative cystoscopy and negative (including atypical) urine cytology
• Positive cystoscopy with biopsy-proven benign or low-grade NMIBC and negative cytology
For intravesical therapies without systemic toxicity, the FDA includes, in the definition of a complete response, negative cystoscopy with malignant urine cytology if cancer is found in the upper tract or prostatic urethra and random bladder biopsies are negative.
Intravesical instillation does not deliver the investigational drug to the upper tract or prostatic urethra. Therefore, the development of disease in these areas cannot be attributed to a lack of activity of the investigational drug. Thus, sponsors can consider patients with new malignant lesions of the upper tract or prostatic urethra who have received intravesical therapy to have achieved a complete response in the primary analysis. However, sponsors should record these lesions and conduct sensitivity analyses in which these patients are not considered to have achieved a complete response.
Systemic therapies are expected to have a treatment effect throughout the urinary tract. Therefore, a patient who received systemic therapy cannot be considered to have a complete response if the patient has a malignant lesion(s) in the upper tract or prostatic urethra.
For the purposes of determining the duration of a complete response, the FDA defines a recurrence as findings on follow-up that no longer meet the above definition for a complete response.
The protocol should provide a plan for the evaluation of patients with suspicious urine cytology. Suspicious cytology does not include the presence of atypical cells. This plan should specify how a suspicious urine cytology will affect the initial definition of complete response and the duration of complete response. For example, the plan may include repeat cytologies or random bladder biopsies.
Regardless of the prespecified plan, all investigators should evaluate suspicious urine cytology in the same manner. The goal of therapy in patients with BCG-unresponsive NMIBC is to avoid cystectomy.”
Theralase®’s Study II treats patients with an intravesical study drug activated by an intravesical study device. In accordance with the FDA Guidance to Industry1, patients enrolled and provided the primary study treatment, where the source of the positive urine cytology has not been identified (i.e.: upper tract or prostatic urethra Urothelial Cell Carcinoma (“UCC”)) and confirmatory bladder biopsies were negative, Theralase® has reclassified these patients from Indeterminate Response (“IR”) to Complete Response (“CR”).
For patients, who have been enrolled and provided the primary study treatment in Study II, that have been diagnosed as IR and do not have confirmatory negative bladder biopsies (confirming that the source of the UCC is not from the bladder wall), then these patients have remained classified as IR, until additional clinical assessments can be completed by the PIs to prove or disprove a diagnosis of CR.
As a result, Theralase® is providing an update to Study II’s interim clinical study data analysis, where some patients have been reclassified from IR to CR on certain assessment days.
In accordance with the FDA Guidance to Industry1, Theralase® will conduct sensitivity analyses, in which these IR patients are considered not to have achieved a CR, as a part of the final clinical report.
Study II objectives are as follows:
Primary:
Efficacy – evaluated by CR at any point in time patients diagnosed with CIS (with or without resected papillary Ta/T1 disease)
CR is defined as at least one of the following:
- Negative cystoscopy and negative (including atypical) urine cytology
- Positive cystoscopy with biopsy-proven benign or low-grade NMIBC and negative cytology
- Negative cystoscopy with malignant urine cytology, if urothelial cancer is present in the upper tract or prostatic urethra and random bladder biopsies are negative
Secondary:
Duration of CR – evaluated as sustainability of CR at 12 months post initial CR.
Tertiary:
Safety – evaluated by the incidence and severity of Adverse Events (“AEs”) directly related to the Study Drug and/or Study Device, Grade 4 or higher that do not resolve within 450 days post treatment (where: Grade 1 = Mild, Grade 2 = Moderate, Grade 3 = Severe, Grade 4 = Life-threatening or disabling, Grade 5 = Death)
Note:
- For patients to be included in the statistical clinical analysis they must be enrolled in Study II, provided the primary Study Treatment and evaluated by a PI at the 90 day assessment visit (cystoscopy and urine cytology)
- One patient passed away prior to their 90 day assessment and is therefore not included in the statistical analysis.
- Evaluable Patients are defined as patients, who have been evaluated by a PI and thus excludes a patient’s clinical data at specific assessment days, if that clinical data is pending.
- Five patients have been enrolled and provided the primary Study Treatment but have not been evaluated at their 90 day assessment; therefore, 52 patients are considered Evaluable Patients at 90 days, with 37 patients considered Evaluable Patients at 450 days.
- The data analysis presented below, should be read with caution, as the clinical data is interim in its presentation, as Study II is ongoing and new clinical data collected may or may not continue to support the current trends, with significant data still pending.
- Total Responders (CR and IR) are defined as CR + IR.
Latest Interim Analysis of Study II:
In Table 1.0 and 2.0, the latest interim clinical data for Study II is presented. Patients, who have been removed from Study II by the PI are included in the analysis; where, their last reported assessment by a PI, prior to removal from Study II, has been carried forward in a Last Observation Carried Forward (“LOCF”) analysis. This LOCF analysis reduces the number of patients analyzed in Study II to only Evaluable Patients (Patients who have been assessed by a PI (i.e.: excludes pending assessments)).
Table 1.0 – Study II Interim Clinical Data – Evaluated Every 90 Days
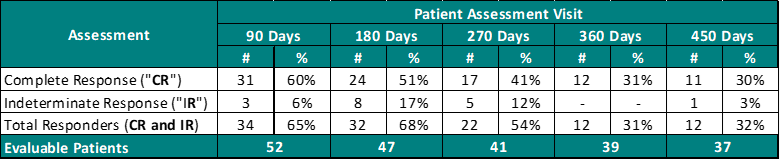
Based on Table 1.0, 60% of Evaluable Patients (Patients evaluated by a PI) achieved a CR at 90 days post primary Study Treatment and 30% achieved a CR at 450 days.
Table 2.0 – Study II Interim Clinical Data – Evaluated According to the Primary, Secondary and Tertiary Objectives
Graphical Representation of the Interim Clinical Data:
The Swimmer’s Plot (Figure 1.0) is a graphical representation of the interim clinical results (n=57) showing a patient’s response to a treatment over time. As can be seen in the plot, significant data is still pending.
Figure 1.0 – Swimmer’s Plot
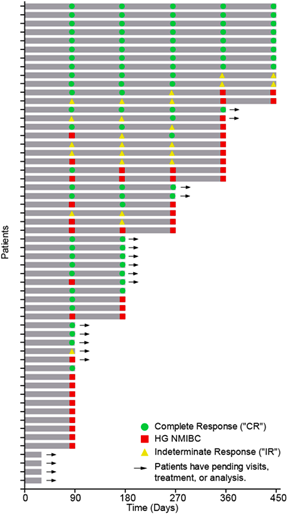
The Swimmer’s Plot illustrates:
11 Evaluable Patients that achieved CR at each assessment date and thus achieved the primary and secondary objectives of Study II for all patients assessed up to 450 days (11/37 = 30%).
34 Evaluable Patients that achieved CR on at least one assessment date and thus achieved the primary objective of Study II (34/52 = 65%)
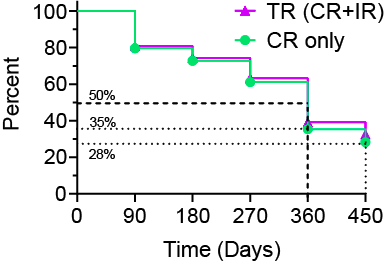
The Kaplan -Meier Curve (Figure 2.0) illustrates the probability of efficacy for the Ruvidar ® Anti-Cancer Therapy (“ACT”) over the study duration, based on the available clinical data.
Figure 2.0 – Kaplan -Meier Curve
About Study II
Study II utilizes the therapeutic dose of RuvidarTM (TLD-1433) (0.70 mg/cm2) activated by the proprietary TLC-3200 Medical Laser System (90 J/cm2). Study II is focused on enrolling and treating approximately 100 to 125 BCG-Unresponsive NMIBC CIS patients in up to 20 clinical study sites located in Canada and the United States.
About TLD-1433 (Ruvidar™)
RuvidarTM is a patented PDC with 12 years of published peer reviewed preclinical research and is currently under investigation in Study II.
The trade name RuvidarTM was selected by the Company, as Ru is the element symbol for Ruthenium, a rare transition metal belonging to the platinum group, which the Theralase ® PDC is based upon, vita is Latin for life and dar is Russian for gift; hence, roughly translated, “Ruthenium, the gift of life”.
About Theralase® Technologies Inc.
Theralase® is a clinical stage pharmaceutical company dedicated to the research and development of light activated compounds and their associated drug formulations with a primary objective of efficacy and a secondary objective of safety in the destruction of various cancers, bacteria and viruses.
Additional information is available at www.theralase.com and www.sedar.com
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward Looking Statement
This news release contains “forward-looking statements” within the meaning of applicable Canadian securities laws. Such statements include, but are not limited to, statements regarding the Company’s proposed development plans with respect to Photo Dynamic Compounds and their drug formulations and a COVID-19 vaccine. Forward looking statements may be identified by the use of the words “may, “should“, “will“, “anticipates“, “believes“, “plans“, “expects“, and similar expressions including statements related to the current expectations of Company’s management for future research, development and commercialization of the Company’s COVID vaccine, including preclinical research, clinical studies and regulatory approvals.
These statements involve significant risks, uncertainties and assumptions; including, the ability of the Company to: adequately fund, and secure the requisite regulatory approvals to successfully complete clinical studies for a COVID-19 vaccine in a timely fashion and implement its development plans. Other risks include: the ability of the Company to successfully commercialize its drug formulations, the risk that access to sufficient capital to fund the Company’s operations may not be available or may not be available on terms that are commercially favorable to the Company, the risk that the Company’s drug formulations may not be effective against the diseases tested in its clinical studies, the risk that the Company’s fails to comply with the term of license agreements with third parties and as a result loses the right to use key intellectual property in its business, the Company’s ability to protect its intellectual property, the timing and success of submission, acceptance and approval of regulatory filings, and the impacts of public health crises, such as COVID-19. Many of these factors that will determine actual results are beyond the Company’s ability to control or predict.
Readers should not unduly rely on these forward- looking statements which are not a guarantee of future performance. There can be no assurance that forward looking statements will prove to be accurate as such forward looking statements involve known and unknown risks, uncertainties and other factors which may cause actual results or future events to differ materially from the forward looking statements.
Although the forward-looking statements contained in the press release are based upon what management currently believes to be reasonable assumptions, the Company cannot assure prospective investors that actual results, performance or achievements will be consistent with these forward-looking statements.
All forward-looking statements are made as of the date hereof and are subject to change. Except as required by law, the Company assumes no obligation to update such statements.
For More Information:
1.866.THE.LASE (843-5273)
416.699.LASE (5273)
www.theralase.com
Kristina Hachey, CPA
Chief Financial Officer
khachey@theralase.com
416.699.LASE (5273) x 224